2 in 1 clinically validated blood pressure monitor with built in ECG to quickly screen for hypertension, AFib, tachycardia and bradycardia at home.

WARNING! BEWARE OF FAKE WEBSITES OR SOCIAL MEDIA ACCOUNTS SELLING COUNTERFEIT OMRON PRODUCTS
Get clinically accurate readings even at home
With OMRON Blood Pressure Monitors
Learn more ↓

Understanding Accuracy in Blood Pressure Monitoring
Being accurate is crucial when it comes to measuring your blood pressure. Accurate measurements mean you can trust the numbers you see. If your devices were not accurate, they could show incorrect readings, which might lead to unnecessary concerns or even improper treatment. To ensure accuracy of measurements, a blood pressure device needs to be clinically validated for accuracy.

Data for Better Health:
When you measure your blood pressure regularly, you provide valuable information for your doctor. They can use this data to adjust your medication and keep it just right for you.
Beat the Nerves:
If your blood pressure tends to rise when you visit the doctor or hospital, don't worry, you're not alone. It's called "white-coat hypertension." Measuring it at home gives a more realistic look at your blood pressure in everyday, relaxed settings.
Set Goals and Succeed:
If you're taking medication or making lifestyle changes to lower your blood pressure, regular and accurate measurements help you set goals and track your progress. Research even tells us that when you keep an eye on your blood pressure, you're more likely to manage high blood pressure.
Let's break it down with a couple of examples: meet Peter and Lucy. What if both Peter and Lucy would have measured their blood pressure with an OMRON device and with an inaccurate device?

Meet Peter, he is 48 years old and preparing for his upcoming appointment with his doctor next Monday. At his last visit, his doctor suggested that he might have high blood pressure and advised him to monitor it for a whole week before the next appointment. Peter took this seriously and measured his blood pressure consistently.
What if Peter had measured his blood pressure with an OMRON device and with an inaccurate device?
-(2).png)

Now, let's introduce Lucy, who is 53 years old and preparing for her doctor's appointment due to suspected high blood pressure. Every time she gets measured at the doctor's office her blood pressure is high. Her doctor recommended that she monitor her blood pressure for three days leading up to her next visit. Lucy followed the instructions and measured her blood pressure on Tuesday, Friday, and Sunday.
What if Lucy had measured her BP with an OMRON device and with an inaccurate device?
.png)
Accurate measurements are crucial for both Peter and Lucy. If their devices were not accurate, they could receive incorrect readings, which might lead to unnecessary concerns or even improper treatment. To ensure accuracy of measurements, a blood pressure monitor needs to be clinically validated for accuracy.
Did you know that all OMRON blood pressure monitors are clinically validated and meet the highest standard of precision and reliability?
It is not enough to just say we are accurate, we must prove it. Which is why we make sure that every model of our OMRON blood pressure monitors meets the highest standards of precision and reliability. This means that our products have been clinically validated. A blood pressure monitor validation study is a clinical study to verify the accuracy of a blood pressure monitor.
Clinical validation offers peace of mind to you and your physician. They can rest assured that you, their patient, are monitoring their health with the same reliability as the medical devices used in their examination rooms.
You probably have seen the CE-mark before. What do you need to know about approved and validated devices in order to make the right decision? In short, if a device has a CE mark, this does not automatically mean it is also clinically validated for accuracy. This means CE-mark devices still can be inaccurate.
![]()
Validated for accuracy per validation protocol
Validation study performed
Not mandatory for selling on EU market

Conformité Européene (CE)
EU mandatory conformity marking for regulating the goods sold within the European Economic Area (EEA) since 1985
Manufacturer has checked that these products meet EU safety, health or environmental requirements
Mandatory when selling on EU market
90% of products in the market are not clinically validated*, and 100% of OMRON blood pressure devices that are sold in Europe are clinically validated for accuracy.
Basic validations are performed on a group of subjects that is representative of an "average population without any diagnosed pre-existing conditions". Special validations are done within a specific group, for instance in the diabetic and pregnancy populations.
Our validated products


Meeting the Highest Standard of Precision and Reliability
The protocol for these validation studies is established by a collaboration of the International Standards Organization (ISO), European Society of Hypertension (ESH), the British and Irish Hypertension Society (BIHS) and the Advanced Association of Medical Instruments (AAMI). STRIDE BP is an international scientific non-profit organization that lists validated blood pressure monitors. STRIDE BP was founded by hypertension experts to improve the accuracy of blood pressure measurement and the diagnosis and management of hypertension.

2 in 1 clinically validated blood pressure monitor with built in ECG to quickly screen for hypertension, AFib, tachycardia and bradycardia at home.

Clinically validated wireless upper arm blood pressure monitor with Intelli Wrap Cuff for easy, accurate readings in any arm position.

Clinically validated smart upper arm blood pressure monitor with Intelli Wrap Cuff, Bluetooth syncing and optional AFib screening for clear, reliable home readings.

Smart, clinically validated upper arm blood pressure monitor with Intelli Wrap Cuff, Bluetooth syncing and AFib detection for simple, accurate home monitoring.

Reliable, easy to use upper arm blood pressure monitor with Intelli Wrap Cuff for accurate home readings and improved measurement consistency.

Smart, clinically validated upper arm blood pressure monitor with Intelli Wrap Cuff, Bluetooth syncing and irregular heartbeat detection for easy home monitoring.

Smart, clinically validated upper arm blood pressure monitor with Intelli Wrap Cuff, Bluetooth syncing and irregular heartbeat detection for easy home tracking.

Clinically validated upper arm blood pressure monitor with Intelli Wrap Cuff, cuff wrap guide and irregular heartbeat detection for easy, accurate home readings.

Easy, clinically validated upper arm blood pressure monitor with Intellisense technology, Easy Cuff and irregular heartbeat detection for dependable home checks.
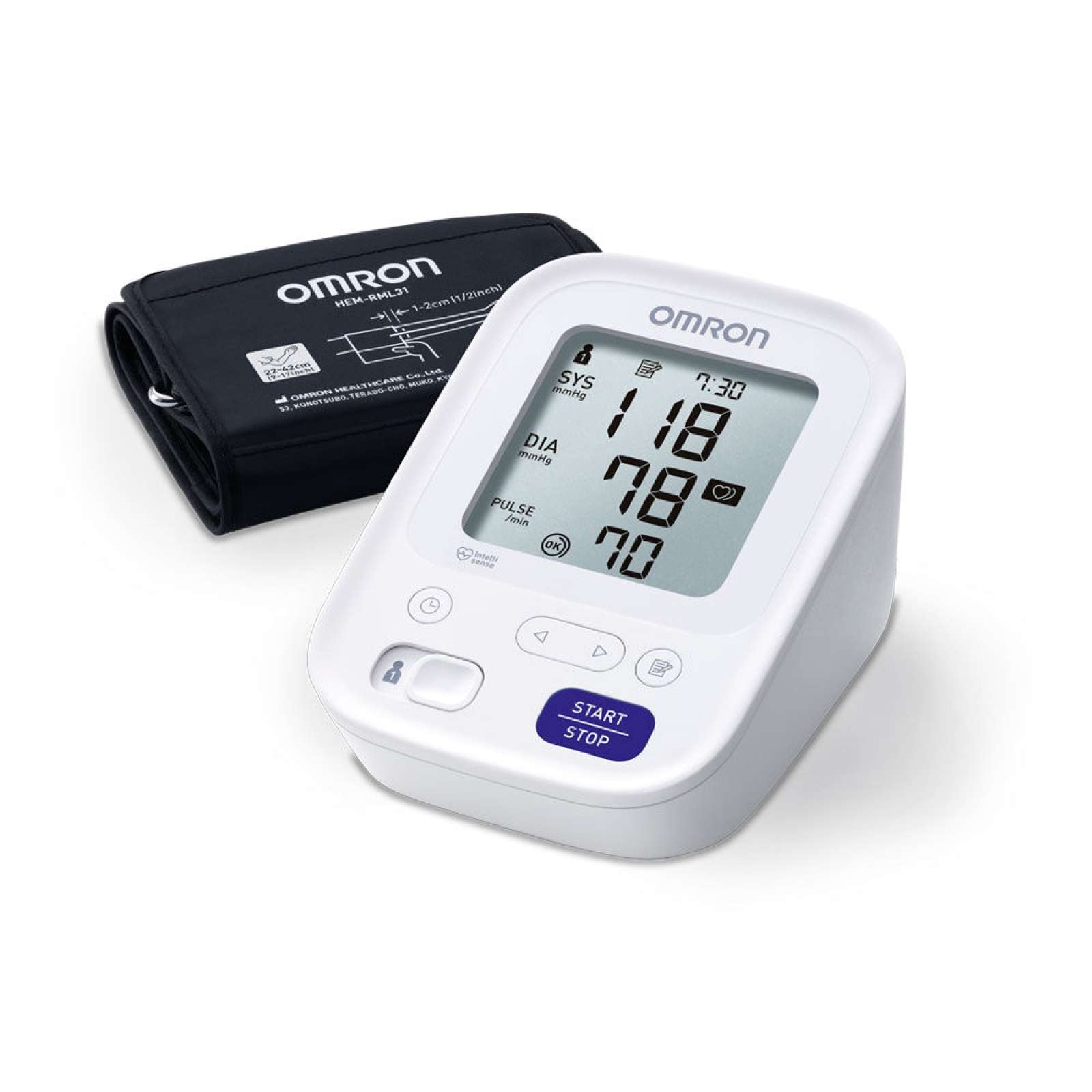
Smart, clinically validated upper arm blood pressure monitor with large 22–42 cm cuff, Intellisense technology and Bluetooth syncing for easy home tracking.

Simple, clinically validated upper arm blood pressure monitor with Intellisense technology and irregular heartbeat detection for reliable everyday readings.

Simple, clinically validated upper arm blood pressure monitor with Intellisense technology, cuff wrap guide and last reading memory for reliable everyday checks.

First at home wrist monitor designed to measure nighttime blood pressure quietly and comfortably for detecting nocturnal hypertension.

Silent, clinically validated wrist cuff blood pressure monitor with positioning sensor and Bluetooth syncing, ideal for accurate monitoring on the go.

Compact, clinically validated wrist cuff blood pressure monitor with Bluetooth syncing for simple, accurate, on the go heart health tracking.

Simple, fully automatic wrist blood pressure monitor with Intellisense technology and irregular heartbeat detection for reliable everyday checks.

Clinical validation can be defined as demonstrating the clinical safety, effectiveness, and efficacy of a diagnosis, treatment, or device. Clinical validation is the process of testing and evaluating by an independent organization to meet clinical standards and meeting the requirements set out by international organizations such as Association for the Advancement of Medical Instrumentation Standard (AAMI) and European Society for Hypertension (ESH).
A clinically validated blood pressure monitor is a device that has been tested in real-world medical settings and functions safely and effectively for its intended use. The testing involves assessing and verifying that the device performs as expected and delivers the intended benefits without causing harm to patients. All OMRON blood pressure monitors are clinically validated.
References:
*= Mancia G, et al. 2023 ESH Guidelines for the management of arterial hypertension. J Hypertens. 2023 Dec 1;41(12):1874-2071.
**= Naseri MW, Esmat HA, Bahee MD. Prevalence of hypertension in Type-2 diabetes mellitus. Ann Med Surg (Lond). 2022 May 14;78:103758.
***= Topouchian J. et al. Clinical accuracy of the Omron M3 Comfort® and the Omron Evolv® for self-blood pressure measurements in pregnancy and pre-eclampsia - validation according to the Universal Standard Protocol. Vasc Health Risk Manag. 2018 Aug 31;14:189-197.
Approval code: OHEAPP-357